-
Use Cases
-
Resources
-
Pricing
....
Events
John Dalton
1808
% complete
Chemists hadn’t yet fully grasped the nature of atoms, as described in the atomic theory proposed by English schoolteacher John Dalton

1860
1860
% complete
Atomic weights had been well enough understood and measured for deeper insights to emerge
John Newlands
1865
% complete
Noticed that arranging the known elements in order of increasing atomic weight produced a recurrence of chemical properties every eighth element
Bohr
1869 - 1922
% complete
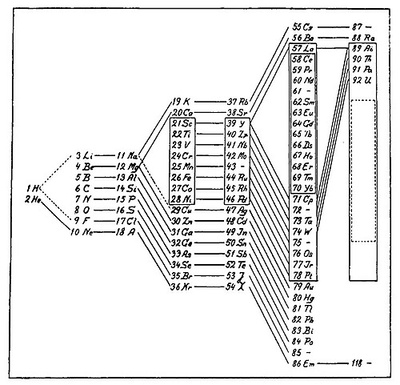
Bohr’s table added elements discovered -1869
Reated his own version of the table- 1922
Dmitri Mendeleev
1869
% complete
he published his firts timeline, this arrangement placed elements with similar properties into horizontal rows.
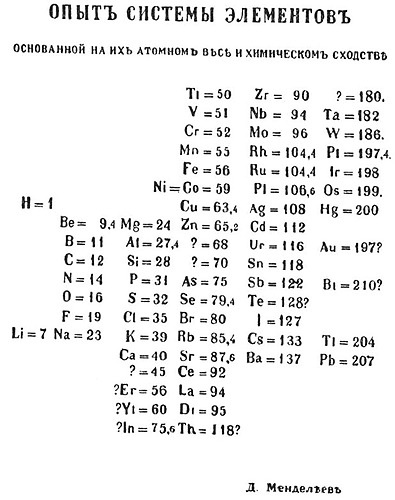
Mendeleev
1869
% complete
His realized that some groups of similar elements showed a regular increase in atomic weights; other elements with roughly equal atomic weights shared common properties.
Johannes Wislicenus
1888
% complete
Declared that the periodicity of the elements’ properties when arranged by weight indicated that atoms are composed of regular arrangements of smaller particles
Mendeleev
1890
% complete
Chemists widely recognized his law as a landmark in chemical knowledge
1907
1907
% complete
Mendeleev’s death
