-
Use Cases
-
Resources
-
Pricing
history of the atom - eva
Events
Democritus
460 BC
% complete
Democritus was born in Greece in 460BC. he then created his model which stated that matter consists of invisible particles called atoms and a void. His atomic model was solid, and stated all atoms differ in size, shape, mass, position and arrangement, with a void exists between them.
http://thehistoryoftheatom.weebly.com/democritus.html

Dalton
1803
% complete
dalton was born in England on the 6th of september 1766. John Dalton proposed that all matter is composed of very small things which he called atoms.
https://www.britannica.com › video › John-Dalton-development-atomic-theory

jj thomson
1897
% complete
jj thomson was born on the 18th december 1856 in britain. jj thomson discovered the electron in 1897 his model was cloud of positive charge filled with atoms and electrons this was called the plum pudding model.
https://www.youtube.com/watch?v=lLwnACfo7hY

Rutherford
1911
% complete
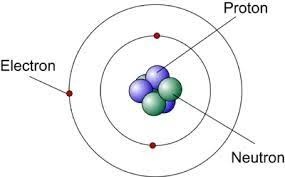
Rutherford was born on the 30th of august 1871 in new Zealand. Rutherford's model shows that an atom is mostly empty space, with electrons orbiting a fixed, positively charged nucleus in set, predictable paths.
https://study.com/academy/lesson/rutherford-model-of-the-atom-definition-diagram-quiz.html

Bohr
1913
% complete
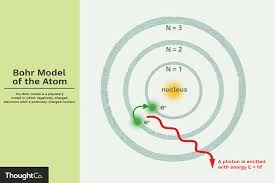
bohr was born on the 7th of october 1885 in Denmark. Niels Bohr proposed a theory for the hydrogen atom based on quantum theory that energy is transferred only in certain well defined quantities. Electrons should move around the nucleus but only in prescribed orbits. When jumping from one orbit to another with lower energy, a light quantum is emitted.
https://www.youtube.com/watch?v=fm2C0ovz-3M
chadwick
1932
% complete
chadwick was born on the 20th of october 1891 in britain. james chadwick discovered the neutron then created a model using rutherfords model interpreting the neutron.
https://www.youtube.com/watch?v=2bNdMzbIuzw