-
Use Cases
-
Resources
-
Pricing
History of the Atom cont.
Events
Democritus: First on atomic timeline
442 BC
% complete
To show scale
Real information is in timeline one
Ernest Rutherford
1911
% complete
He conducted research in Montreal where he discovered the half life of atoms and alpha/gamma/beta radiation. He won the 1908 Nobel Prize in Chemistry for these studies. In 1911 he conducted an experiment where he fired alpha particles at golf foil and observed the particles bouncing back. He concluded that there was both positive and negative charge in the electron and that most of the atom was empty with most of the mass residing in the the nucleus in the center and electrons orbiting around. He also was the first to transform one element into another when he converted nitrogen atoms to oxygen atoms, discovered the proton, and predicted the existence of the neutron.
Henry Mosely
1913
% complete
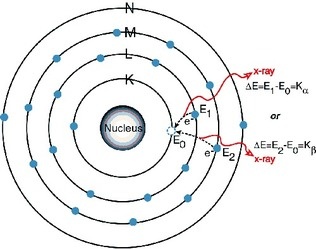
Mosely established that the number of protons determines the atomic number of an element, not mass which was used before. He X-rayed different metals and found the wavelengths created a linear graph that corresponded with atomic numbers. He then reorganized the periodic table and many of the predicted elements turned out to be true.
Niels Bohr
1913
% complete
Bohr developed the Bohr model from the spectral line series emitted by hydrogen. The spectral line series was energy released in wavelengths by electrons as they jumped from one energy level to another on a hydrogen atom. His model was the first to show electrons rotating in different orbits around the nucleus and that the number of electrons in the outer orbit showed properties of the given element. Bohr was given a Nobel Prize in 1922.

Werner Heisenburg
1925
% complete
Heisenburg studied quantum mechanics and quantum theory. At the time the Bohr model was in use, where electrons were describes as being in a a fixed orbit. However, the model only worked with the hydrogen atom. Heisenburg developed the uncertainty principle which stated that the more precisely the position of a particle is known, then the less precisely the momentum of the particle is known. This did not change much of Bohr's model, only that the precise location of the electron cannot be known. Bohr won the Nobel Prize in Physics in 1932.
Erwin Schrödinger
1926
% complete
Schrödinger is known for his equation and more famously the Schrödinger's cat paradox. ( https://whatis.techtarget.com/definition/Schrodingers-cat ) Schrödinger developed a mathematical equation that calculated the possibility of an electron's location known as the quantum mechanical model of the atom. While the Bohr model defined the path of the electron, the quantum mechanical model of the atom predicts the possibility of the atom being in a given location. Schrödinger won the 1933 Nobel Peace Prize in Physics for his equation.
James Chadwick
1932
% complete
Chadwick experimented with beryllium by bombarding it with alpha particles and observed it released a radiation with an identical mass to protons but had no charge. This unknown radiation turned out to be neutrons. It was also discovered that neutrons could directly penetrate the nucleus of an atom. This was a very helpful contribution to the development of the atomic bomb.

Sources
Present
% complete
A photo of a single atom taken in modern day.
https://www.atomicheritage.org/profile/ernest-rutherford
https://www.ducksters.com/biography/scientists/antoine_lavoisier.php
http://www.abcte.org/files/previews/chemistry/s1_p6.html http://web2.uwindsor.ca/courses/physics/high_schools/2005/Brownian_motion/History.html
https://historyoftheatomictheory.weebly.com/aristotle.html
http://www.abcte.org/files/previews/chemistry/s1_p4.html
https://courses.lumenlearning.com/cheminter/chapter/spectral-lines-of-hydrogen/
https://www.atomicheritage.org/profile/james-chadwick
https://www.atomicheritage.org/profile/marie-curie
https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/history-of- atomic-structure/a/daltons-atomic-theory-version-2
http://thehistoryoftheatom.weebly.com/democritus.html
https://www.famousscientists.org/erwin-schrodinger-2/
https://www.famousscientists.org/j-j-thomson/
https://www.famousscientists.org/henry-moseley/
https://web.lemoyne.edu/~giunta/EA/ARISTOTLEann.html
